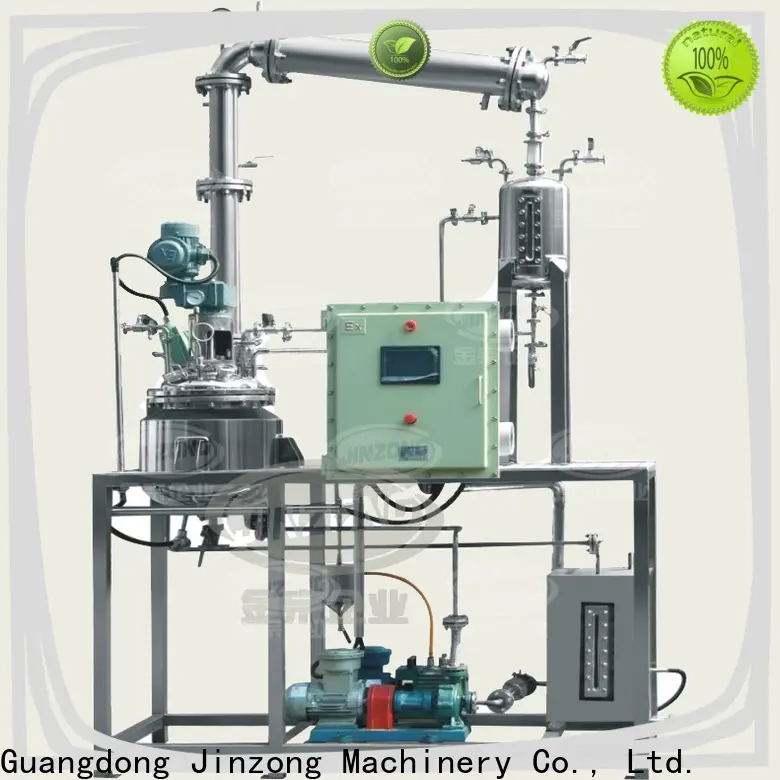
Esterification reaction is an important type of organic chemical reaction, usually referring to the process in which alcohols react with carboxylic acids (or anhydrides) under the catalysis of acids or bases to form esters and water. This reaction is widely used in food processing, chemical industry, medicine, essence, spices, coatings and other industrial production fields.Esterification reaction, as a key link in chemical production, has always been a focus of industry attention in terms of production efficiency and quality control. The reaction process requires precise control of parameters such as temperature, pressure, and stirring speed, providing a stable and efficient reaction environment for esterification reaction. In the actual production of esterification reactions, there are often many problems such as low space utilization, uneven stirring, low reaction efficiency, insufficient utilization of heat energy, and single production conditions.After years of research and technological breakthroughs, Jinzong Enterprise has launched an intelligent esterification reaction production line, which completely solves the above pain points, promotes the digital upgrade of esterification reaction production, helps customers reduce costs and increase efficiency, and creates new quality productivity.1、 Structural compositionThe Jinzong esterification reaction intelligent production line consists of multiple core modules, including an efficient preheating system, intelligent reaction kettle, automated stirring device, high vacuum system, heat recovery system, and intelligent control system.Efficient preheating system: By efficiently preheating the reaction materials, the reaction time of the materials in the reaction kettle is shortened, while avoiding the thermal degradation side reaction of the products and improving the product yield.Intelligent Reactor: Equipped with a built-in heating device and advanced sensors, it can monitor reaction temperature, pressure, and other parameters in real time to ensure precise control of reaction conditions.Automated mixing device: equipped with a high-precision mixer, it can automatically adjust the mixing speed and angle according to the reaction requirements, achieve uniform mixing of materials, and improve product quality.High vacuum system: With vacuum pump as the core, it consists of vacuum measuring device, vacuum pipeline and valve, condensing device, buffer tank, filtering device and control system, etc., creating a high vacuum environment for esterification reaction, improving reaction efficiency and product quality.Thermal energy recovery system: Utilizing advanced heat exchange technology, the waste heat generated during the reaction process is recovered and reused, significantly improving energy utilization efficiency.Intelligent control system: With DCS process control system as the core, coupled with MES formula management system, SIS safety interlock system, ESS emergency shutdown system, it realizes the automation, intelligent monitoring and optimization of the production process.The functions include: automatic recipe management, metering and addition, equipment operation, conveying and packaging, storage and palletizing, equipment operation and monitoring, fault grading alarm, fault interruption handling, equipment load analysis, equipment working condition warning, equipment maintenance guidance, remote uploading and downloading, etc.2、 Functional FeaturesHighly automated: From material preheating, reaction control to product collection, the entire production process has achieved fully automated operation, reducing manual intervention and labor intensity.Precise control: Through advanced sensors and control systems, parameters such as reaction temperature, pressure, and stirring speed can be precisely controlled to ensure the stability and consistency of reaction conditions.High vacuum: accelerates the reaction process, prevents reverse reactions, and improves production efficiency; Remove impurities and by-products to improve the quality and purity of the product; Prevent oxidation and hydrolysis to ensure the stability of reactants and products; Facilitating material transportation and improving safety.Data driven optimization: Utilizing big data analysis and artificial intelligence technology to collect real-time production data, optimize production processes, predict and prevent potential problems, and further improve production efficiency and product quality.Flexible production conditions: reaction parameters and equipment configuration can be quickly adjusted according to the needs of different esterification reactions to meet diverse production requirements.Prefabricated: The production line realizes modular assembly, greatly reducing installation and debugging time. The project production time is advanced by about 60%, providing assistance for customers to quickly seize the market. 3、 Advantages and highlightsSignificantly improving space utilization: adopting compact reaction equipment and vertical arrangement design, greatly reducing the footprint and improving the utilization efficiency of production space.Uniform stirring and high reaction efficiency: The automated stirring device can adjust the stirring method in real time according to the reaction requirements, ensuring thorough mixing of materials and significantly improving reaction efficiency.Energy conservation and consumption reduction: The thermal energy recovery system recovers and reuses the waste heat generated during the reaction process, reducing energy consumption and production costs.Safety and environmental protection: The intelligent control system can monitor various parameters in the production process in real time, detect and handle abnormal situations in a timely manner, and ensure the safety and reliability of the production process.Stable Quality: The application of high vacuum systems, precise control, and data-driven optimization ensure high consistency and stability in product quality, reducing the rate of defective products.Easy to clean: The cleaning component of the stirring blade can scrape off the adsorbed raw materials on the inner wall of the reaction vessel, reducing the difficulty of cleaning for users and facilitating the cleaning of the inner wall of the reaction vessel.4、 Application FieldsThe Jinzong esterification reaction intelligent production line has strong applicability and can be widely used in multiple industries, mainly including the following aspects:Food industry: In the food industry, esterification reactions are used to manufacture food flavorings. For example, the esterification reaction between ethanol and acetic acid can produce ethyl acetate, a compound with an aromatic odor commonly used for seasoning and enhancing the flavor of food.Spice manufacturing: Ester compounds are the main components of many natural and synthetic fragrances. Through esterification reactions, ester fragrances with specific aromas can be synthesized to meet people's needs for different fragrances.Coatings and plastics industry: Some ester compounds have good film-forming and weather resistance, and are therefore used as raw materials for coatings and plastics. Through esterification reactions, ester compounds with specific properties can be synthesized to meet the needs of the coatings and plastics industries.In the pharmaceutical field, esterification reactions are involved in the synthesis of many drug molecules. The Jinzong esterification reaction intelligent production line, with its precise control ability and efficient production efficiency, can provide important support for pharmaceutical production.Organic synthesis: Esterification reaction is one of the important methods for constructing carbon carbon and carbon oxygen bonds in organic synthesis. Through esterification reaction, various ester compounds can be synthesized, which are widely used in pesticide, dye, medicine, essence, spice and other fields.Supported by specialized and innovative enterprises, the quality of service is guaranteedJinzong Enterprise was founded in 1999 and has over 20 years of experience in the production, research and development, design, and installation of food and pharmaceutical equipment. It has a design and marketing service center in Guangzhou and has two production factories and a provincial-level engineering technology research center in Zhaoqing National High tech Zone. Jinzong Enterprise has the qualifications for manufacturing special equipment pressure vessels and installing pressure pipelines (GC2), and is a national high-tech enterprise and a provincial-level specialized, refined, and new enterprise. Jinzong Enterprise has two provincial-level famous brand products, dozens of product patents, software copyrights, and provincial-level high-tech products. It has passed the national intellectual property standard certification ISO9001-2015 international quality system certification, EU CE certification, and five-star after-sales service certification. It has been rated as a "Guangdong Province Contract abiding and Creditworthy Enterprise" by the Guangdong Provincial Administration for Industry and Commerce for many years. With strong technical research and development capabilities and guaranteed service credit, it has customers in more than 50 countries and regions around the world, and has gained widespread recognition and support from more than 2000 large and small enterprises at home and abroad. To do a good job, we must first sharpen our tools. "Jinzong Enterprise adheres to the concept of" quality is like gold, craftsmanship is the master "and provides advanced and automated production lines to manufacturing factories. We welcome friends from home and abroad to visit and guide us.
Read More>>
All Products
- PRODUCT Supplier
-
Quality Pharmaceutical produciton equipment
-
Buy Filling & Package Equipment Supplier
-
Storage Tanks and Vessels Factory
-
Quality Chemical reaction and mixing plant Supplier
-
Customized Paint & Coating Production Equipment
-
Hot 推广不显示 Supplier
-
Packeage equipment Supplier
-
Tube filling and sealing machine For sell
-
Dispersing and mixing tank and vessel Wholesale
-
Best Others Supplier
-
Best Accessories for Machinery
-
Buy RO Pure Water Equipment
-
Best Storage Tanks and Vessels
-
Labeling Machine Wholesale
-
Others Filling & Package Equipment Factory
-
Weighing filling machine Warranty
-
Best Cosmetic/food/pharmaceutical filling machine Bulk
-
Customized Turnkey project for pharmaceutica factory Factory
-
Turnkey project for food factory For sell
-
Hot Food production equipment
-
Hot Others Food and pharmaceutical Production Equipment Exporter
-
Quality Turnkey project for paint&coating factory
-
Top Miller and grinder
-
Laboratory equipment For sell
-
Best Others Paint & Coating Production Equipment Bulk
-
Buy Dissolver and disperser Supplier
-
Pharmaceutical produciton equipment Exporter
-
Certificated PRODUCT For sell
-
Chemical Processing Machine Manufacturer
-
Pharmaceutical and Food Processing Machine For sell
-
Quality Cosmetic Processing Machine
-
Paint and New Energy Processing Machine Exporter
-
Intelligent Production System Company
-
Top Pharmaceutical and Food processing machine
-
Cosmetic processing machine Bulk
-
Quality Chemical Processing Machine Company
-
Paint and New Energy processing machine Wholesale
-
Top Pharmaceutical and Food processing machine Bulk
-
Certificated Intelligent Production System Warranty
- Quality Paint and New Energy processing machine
-
Buy Pharmaceutical processing machine Exporter
-
Customized Cosmetic processing machine
-
Chemical Processing Machine Wholesale
-
Chemical Processing Machine Manufacturer